Rank the Following From Most Acidic to Least Acidic.
Find an answer to your question Rank the following from most to least acidic. -CO -COM -CO-H HOS -CON O A Posted 2 months ago.

Solved Rank The Following Solutions In Order From Most Chegg Com
E CH 3 CH 2.
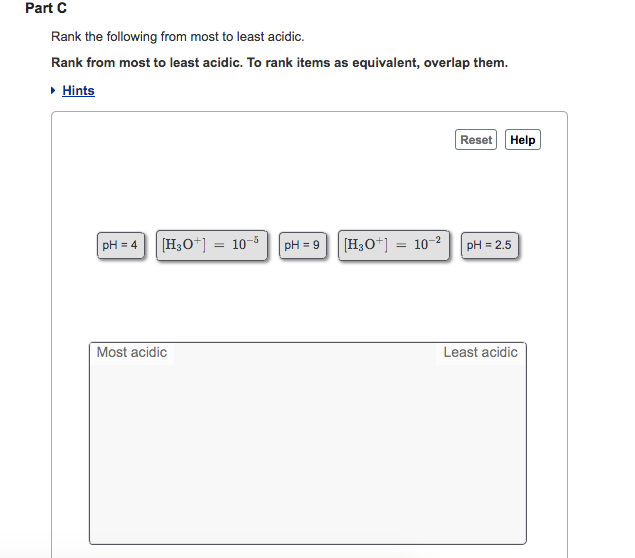
. The answer is. Rank the following from most to least acidic. To rank items as equivalent overlap them.
Rank from most to least acidic. And we can judge these hydrogen acidity based on the stability of their constant basis. Rank from most to least acidic.
The pH expresses the molar concentration of hydronium ions in an aqueous solution on a logarithmic scale. And so we know that resonance will stabilize a base. A h3coh b h3ccoch3 also please check this rank the following from most to least acidic compound.
44 Rank the bold-faced hydrogens for the following compounds from most acidic to least acidic. Solved Rank the following from most to least acidic. To rank items as equivalent overlap them.
Since presence of NO 2. Solution X pH 650 A Z YX B YXz Solution Y pOH 1030 CYZX D X YZ Solution Z OH- 42 x 10-9 M. Memorize flashcards and build a practice test to quiz yourself before your exam.
Part C pH -log H3O Rank the following from most to least acidic. Click hereto get an answer to your question Rank the following compounds in order from most acidic to least acidic. Question 15 Rank The Following Compounds In Order Of Increasing Acidity From Least Addic To Most Acidic.
Hence the order of acid strength is 3142. Ah2so4h3cco2hh20 bh3cch2nh2h3cch2ohh3cco2h Science Chemistry according to the reaction below. Rank these items from most acidic to least acidic H3O102 pH 3 pH 5 H3O106 pH 14 - The 10-6 M H3O solution has a pH of 6 and the 10-2 M H3O solution has a pH of 2.
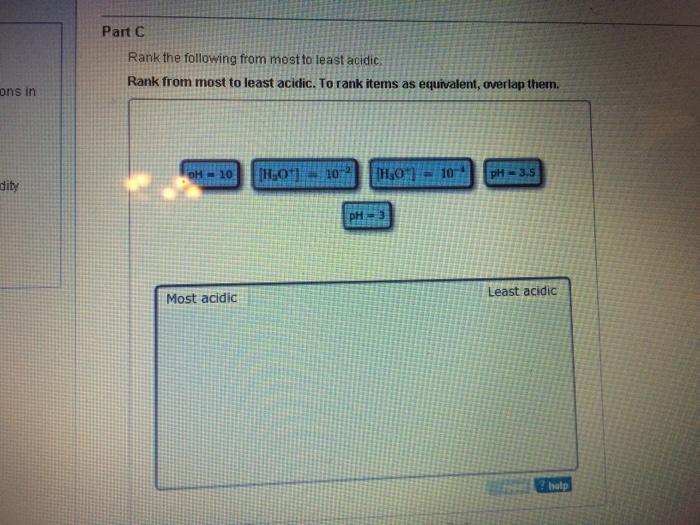
I II III IV V H H O H O H O H O CH 3 H Answer. Rank the following from most acidic to least acidic. View Available Hints Reset Help H3O 10 x 10-6 mol L 1 pH 5 pH 14 pH 3 3D H30 10 x 10 2 mol L- Most acidic Least acidic.
Rank these items from most acidic to least acidic. The weakest acid in the table is. Lets keep an eye on the content below.
Rank the bold-faced hydrogens for the following compounds from most acidic to least acidic. The solutions are ranked from most acidic pH 2 to most basic pH 14. Rank the following from most acidic to least acidic.
Help pH 3 pH 14 pH 5 IT H2O4 102 H2O. B it can dissolve in water. Rank from most to least acidic.
4 NO 3 O2 2 H2O 4 HNO3 a 0001 g of NO dissolves into 1 liter of rainwater and all of it transforms into nitric acid. Rank the equivalence point EP of the following titrations from most acidic to least acidic in the following order. нн The following solution is suggested to handle the subject 4.
C it is a hydroxide donor. Rank the following from most to least acidic. Rank the following from most to least acidic.
Rank the following in order of most acidic to least acidic. This question asked us to rank the indicated hydrogen is in each of these compounds from most acidic to least acidic. Rank the bold-faced hydrogens for the following compounds from most acidic to least acidic.
O a 20 M C H N. PH 31 pH 5 11130-11 10-least acidic. Chemistry questions and answers.
Rank the following groups of compounds from most acidic 1 to least acidic 4. Rank the equivalence point EP of the following titrations from most acidic to least acidic in the following order. Group in para position causes more electron-withdrawing effect than that in meta position 1 will be less acidic than 3.
Join Login Class 12 Chemistry Alcohols Phenols and Ethers Chemical Reactions of Alcohols and Phenols Rank the following compounds in order fr. Which of the acids has the weakest conjugate base. Rank the following from most to least acidic.
To rank items as equivalent overlap them. Rank these items from most acidic to least acidic. III I IV V II 45 Select the strongest base.
To rank items as equivalent overlap them. PH3 pH35 H3O 10-2 pH10 H3O10-4. H3O10-2 ph3 ph5 H3O.
Rank the following from most to least acidic. Solution for Rank the following in order of most acidic to least acidic. Rank the following from most to least acidic.
Further presence of electron withdrawing group and its stronger effect stabilizes the phenoxide more. Solution X pH 850 3D A Z X Y B Y X Z Solution Y pOH 430 C Y Z X. Chemistry questions and answers.
So the contact base would be an an eye on if the loan hair on that carbon nestle carbon on and the more residents. Start studying the Chemistry Chapter 14 Study flashcards containing study terms like NaOH is a Brønsted-Lowry base because A it is a polar molecule. Most acidic EP 1 to the least acidic EP 5 Which titration will be ranked 4.
D it is a proton acceptor Which acid-base definition classifies an acid as an electron-pair acceptor. Rank from most to least acidic. To rank items as equivalent overlap them.
Hence 2 will be least acidic. Rank from most to least acidic. Rank the following in order of most.
View Available Hints Reset Help H3O HO 10-pH 35lpH 10 H3O 10-2 pH 3 Most acidic Least acidic. In m-nitrophenol the inductive effect of the electron-withdrawing nitro group helps to stabilize the. A OH b RC C c NH 2 d CH 2 CH e CH 3 CH 2 Answer.
View Available Hints most acidic pH 31. Solve Study Textbooks Guides. To calculate pH and use it as a measure of acidity.

Solved Part C Rank The Following From Most To Least Acidic Chegg Com
Solved Rank The Following From Most To Least Acidic Rank Chegg Com

No comments for "Rank the Following From Most Acidic to Least Acidic."
Post a Comment